Glycobiology and Extracellular Matrices.Ubiquitin is a small 8-kd stress protein that may facilitate targeting and removal of other proteins denatured during the stress event. The 20-kd protein, Hsp20, found in vascular smooth muscle, is a substrate for protein kinase and probably has a role in the maintenance of vascular tone and vessel wall remodeling. Many of the small stress proteins that are present in the cytosolic compartment may be important in cardiovascular biology. Mitogen-activated protein kinases are involved in the intracellular signaling cascade and are activated during ischemia-reperfusion. Phosphorylation of Hsp25 occurs via the mitogen-activated protein kinases. Physiologic stress increases the phosphorylation of Hsp27. Hsp25/27 influences the cell cytoskeleton (actin polymerization) and may be involved in cell migration. There is no direct evidence that Hsp32 functions as a chaperone, but its overexpression during stress events indicates that it may function in this fashion. Hsp32 is induced by sheer stress and may mediate nitric oxide–dependent platelet inhibition and vasodilatation. Hemeoxygenase (Hsp32), a rate-limiting enzyme in the degradation of Heme, is stress induced and is abundant in myocardial cells. These factors may make aging tissues more susceptible to oxidative stress injury. It appears that there is decreased expression of stress protein genes and decreased activity of HSF-1 during aging. These proteins now are being implicated in the aging process. In addition to being involved in specific disease processes, the stress proteins may play a key regulatory role in cell death pathways (apoptosis) that involve DNA and protein synthesis. Hsp60/65 is found in high concentrations in human arteriosclerotic lesions, and there is a correlation between anti-Hsp60/65 antibodies and atherosclerosis. Experimentally, arteriosclerotic lesions can be induced by immunization with Hsp60/65. There is a growing body of evidence that some stress proteins may be associated with atherosclerosis. 1).Ībnormal levels of stress proteins have been found in a number of disorders, including atherosclerosis, congestive heart failure, fever, infection, aging, Alzheimer's disease, malignant diseases, and autoimmune disorders. 
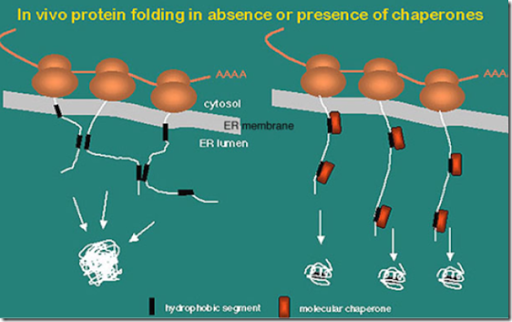
In fact, they ensure that newly formed polypeptides proceed correctly through folding and unfolding to eventually achieve a functional shape (Fig. Stress proteins are critically important because they appear to be necessary in the critical step of three-dimensional folding of some newly formed proteins within the cell. Consequently, “stress protein” is the preferred term. Thus, the term “heat shock protein” is a misnomer because many agents other than heat induce the expression of the heat shock protein gene. However, the genes are now known to be induced by a wide variety of environmental or metabolic stresses that include the following: anoxia, ischemia, heavy metal ions, ethanol, nicotine, surgical stress, and viral agents. This family of genes originally was named because of their expression after exposure to heat. The DNA sequence that makes up this family of genes is highly conserved across species. Heat shock proteins originally were discovered when it was observed that heat shock produced chromosomal puffs in the salivary glands of fruit flies ( Drosophilia). Many types of stress, including heat, induce expression of a family of genes known as the heat shock protein genes. Heat shock proteins are ubiquitous proteins found in the cells of all studied organisms. Journal of Vascular Surgery Cases, Innovations and Techniques.Journal of Vascular Surgery: Venous and Lymphatic. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
